 The Dosage layer, the final level of the PSP hierarchy, imposes a periodic Dosage ON and Dosage OFF mechanism onto the underlying Pulse Pattern and Train layers. It defines sequences of stimulation “trains” during which the underlying Pulse Pattern is repeated. The Train layer modulates the Pulse Pattern layer and is the next level of the PSP hierarchy. The Pulse Pattern layer operates with the finest granularity of the three layers and comprises repeating periods of stimulation and quiescence. Each of these layers is independently configurable to offer and achieve optimal pain relief for each unique patient. The PSP family of waveforms is composite signals created by layering specific temporal patterns (Pulse Patterns, Trains, and Dosages) in a hierarchical structure. This method may drive the simultaneous benefits of multiple, different therapies with a single pattern. The novel PSP used here is a proprietary, unique, layered therapeutic approach that allows each stimulation layer to be individually adjusted to potentially tap into a distinct mechanism of action. 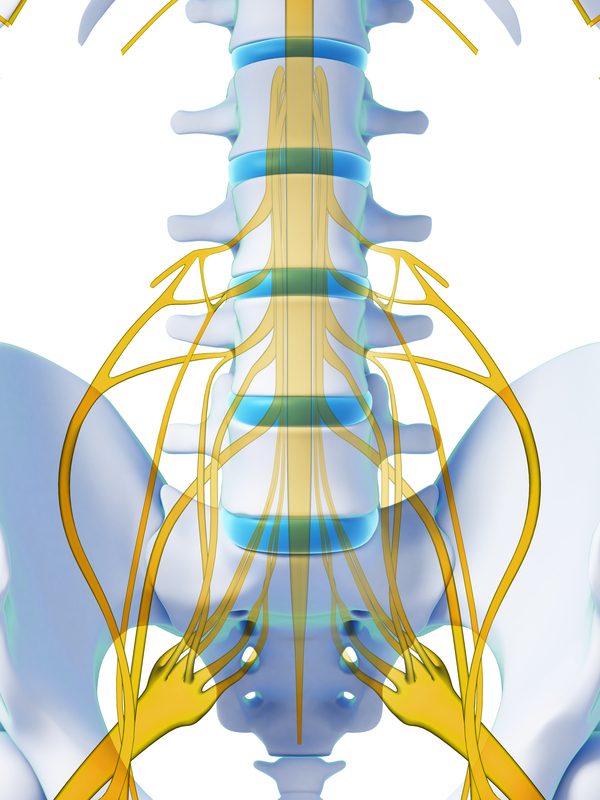
Among the options included was a novel, paresthesia-independent, PSP with the amplitude optimized for each subject. Multiple therapy options were offered to each subject. Programming of each implanted device was performed by clinical personnel from the study sponsor, under the direction of a study physician. The system was programmed for optimal pain relief. 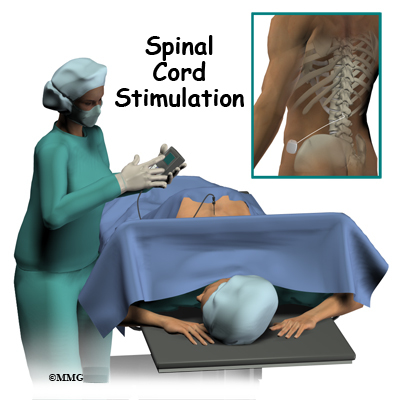
The brief pain inventory (BPI) was used to evaluate the severity of pain and its impact on functioning. Baseline assessments were completed including pain ratings for both leg and/or back pain using the 11-point NRS (0 = no pain 10 = worst possible pain) and the Visual Analog Scale (VAS) of 0 to 10 cm (0 = no pain 10 = worst pain imaginable). The key exclusion criteria included 1) previously failed SCS 2) sensitivity to adhesives worn on the skin 3) > 120 mg morphine equivalents per 24 hours and 4) mechanical spine instability. 
The key inclusion criteria included 1) chronic intractable pain of the leg(s) and/or back, predominantly neuropathic (based on investigator’s clinical judgment, physical examination, and presenting pain symptoms) 2) baseline Numerical Rating Scale (NRS) pain diary score of ≥ 6 3) diagnosis of failed back surgery syndrome and 4) ability to appropriately place an adhesive disc on one’s back. Subjects recruited from six comprehensive pain centers in Australia who met the inclusion/exclusion criteria and signed informed consent were enrolled in the study. This prospective, open-label feasibility study was conducted to obtain clinical data on the safety and efficacy of a neurostimulation system for SCS (Nalu Medical, Inc, Carlsbad, CA). In addition, we hypothesize that the pulsed stimulation pattern (PSP) waveform provides pain relief that is comparable with other commercially available waveforms. The design hypothesis for this novel, battery-free, miniaturized IPG is to provide a comfortable system to patients that decreases pain at the implant site owing to its small size (< 1.5 cm 3) compared with historical SCS systems with larger implants. In addition, the unique microchip design of the system allows software upgrades over time for additional capabilities, parameters, and therapy options. This IPG (< 1.5 cm 3) delivers pulses to one or two percutaneous leads (up to eight contacts each) placed in the epidural space for SCS or in proximity to a nerve for peripheral nerve stimulation and can support a broad menu of therapy options, including novel waveforms. The device is not cleared to treat pain in the craniofacial region. The FDA has cleared the system for two indications: 1) SCS as the sole mitigating agent, or as an adjunct to other modes of therapy used in a multidisciplinary approach for chronic, intractable pain of the trunk and/or limbs, including unilateral or bilateral pain and 2) peripheral nerve stimulation for pain management in adults who have severe, intractable chronic pain of peripheral nerve origin, as the sole mitigating agent or as an adjunct to other modes of therapy used in a multidisciplinary approach. Recently, a battery-free, miniaturized IPG, < 1.5 cm 3 volume, has been developed that is powered by a small, externally worn battery pack (Therapy Disc ). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
